Composition for Treating Neurodegenerative Diseases
Neuroshield is a new molecule that can penetrate the brain and reduce inflammation in mice models with Parkinson’s Disease. It has the potential to slow down the progression of the disease without adverse effects thanks to its unique mode of action.
BACKGROUND
With increasing life expectancy, over time incidence of neurodegenerative diseases, nervous system dysfunction and other age-related syndromes has accelerated. Current therapies for neurodegenerative diseases only alleviate physical complications, being unable to act on mechanisms underlying neurodegeneration. The determinant role of misfolded protein aggregation, oxidative stress, DNA damage, uncontrolled signaling, have been seen as novel opportunities to stop or inhibit disease progression. However, currently, neuroinflammation is gaining insights as prominent therapeutic approach. Indeed, growing evidence has demonstrated neuroinflammation as a key process in the pathogenesis of major neurodegenerative diseases.
Neuroinflammation is controlled by microglial cells, the innate immune cells of the central nervous system (CNS), that are considered the “sentinels” of CNS. Under transient damage, microglial cells protect their surrounding microenvironment and maintain the normal functions of CNS, by playing immune resolving, anti-inflammatory functions and supporting cell renewal by secreting trophic factors. Vice versa, under intensive pathological acute or chronic activation, microglia cells become neurotoxic and secrete a plethora of pro-inflammatory cytokines, prostaglandins and reactive oxygen (ROS) and nitrogen (NOS) species, impairing thus neuronal activity, disturbing normal neurotransmitter function and irreversible leading to tissue loss.
TECHNOLOGY OVERVIEW
The present invention refers to a composition of (poly)phenol metabolite-based compounds. With a unique mode-of-action not toxic for the cells, our invention targets neuroinflammation – a key driver for neurodegeneration. Thus, our invention is applicable for preventing and/or treating neurodegenerative diseases, through radically different approach face to competitors.
Main features:
- The composition decreases, by approximately 50%, the release of pro-inflammatory cytokines mainly TNF-α, IL-6, IL-10, IL-1, IL-12, IL-18, IL23, CCL17, CCL22 and CXCL1, in microglial cells;
- The composition decrease of translocation of transcription factors mainly NF-kB, upon inflammatory insult, to control levels, in microglial cells;
- Decrease TLR4 expression by approximately half, after 6 hours of pre-incubation, in microglia cells;
- Increase of cell viability and ATP levels by approximately 30% in MPP+ exposed dopaminergic neurons.
Deriving from resorcinol, a broad portfolio of active compounds can be engineered into molecules highly active at low concentrations (lower toxicity/lower side-effects) and accessible to blood brain barrier (BBB). These capabilities dramatically increasing the diversity of potential targets, including but not limited to: Alzheimer’s disease, Parkinson’s diseases, amyotrophic lateral sclerosis, muscular dystrophy, multiple sclerosis, brain cancer, epilepsy.
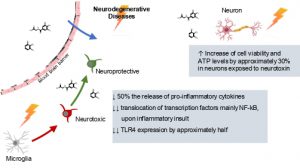
Figure 1: Graphical abstract representing the protective effect of the composition towards neurodegenerative hallmarks: reduction neuroinflammation in microglia and increasing viability and functionality of neurons.
STAGE OF DEVELOPMENT
Technology Readiness Level (TRL): 3 – Experimental Proof of Concept
Recent Developments and Applications
To demonstrate the effectiveness of the technology, the team conducted small preliminary pilot study exploiting direct administration on a mice model of (neuro)inflammation using lipopolysaccharide insult. We were able to validate the capability of our resorcinol-based compounds to reduce the presence of inflammatory cytokines both in blood and the brain. Furthermore, we observe the decrease in other inflammatory markers such as NF-kB and TLR4 as previously denoted in vitro in our patent application. The researchers continuously working on this project and generating more evidence on the potential future applications of our technology.
BENEFITS
Our resorcinol-based solution is effective at very low concentrations, which are identical to achievable circulating blood concentrations. Thus, the potential of their use as novel non-toxic pharma compounds to target inflammation-based pathologies is tremendous.
APPLICATIONS
Resorcinol-based, small (poly)phenol metabolites has the capability to enter in circulation at what we proved to be effective concentrations for treating neuroinflammation. Importantly, these small molecules have also the ability to reach the brain. Thus, nutritional and/or pharmacological approaches to deliver these molecules can be developed aiming to:
- Modulate neuroinflammation in the innate immune cells of the brain (microglial cells);
- Increase cell viability in neuronal cells exposed to a Parkinson’s inducing toxin (MPP+).
The preventive profile by measuring different doses and time of incubation in microglial cells has already been characterized in vitro. Meanwhile the mechanisms by which these molecules are being elucidated. Furthermore, this substance has been tested in a preventive manner in neuronal cells treated with Parkinson’s inducing toxin. The results showed the capability of the metabolites to increase neuronal cell viability, thus showing they are able to protect neurons from dying. The full mechanism behind this protection is under study. For this model we are still awaiting further animal validations in the future.
Overall, the gathered evidences show the potential of these small phenolic molecules to be used in a preventive manner for the deterrence of neuroinflammation and neurodegeneration and potentially be used in patients with these complications.
INTELLECTUAL PROPERTY
- WO2023156983
- EP4230201A1
- PT11701 (Priority date: 21/02/2022).
FURTHER DETAILS
Figueira I, Garcia G, Pimpão RC, Terrasso AP, Costa I, Almeida AF, Tavares L, Pais TF, Pinto P, Ventura MR, Filipe A, McDougall GJ, Stewart D, Kim KS, Palmela I, Brites D, Brito MA, Brito C, Santos CN. Polyphenols journey through blood-brain barrier towards neuronal protection. Sci Rep. 2017 Sep 13;7(1):11456. doi: 10.1038/s41598-017-11512-6. Erratum in: Sci Rep. 2021 Aug 18;11(1):17112. PMID: 28904352; PMCID: PMC5597593.
OPPORTUNITY
- Available for exclusive licensing and/or seeking co-development partners for the realization of clinical trial accessing the efficacy of the drug in humans for future implementations in the clinic as pharmacological compounds and/or nutritional approach.
- Available for exclusive and non-exclusive licensing as lead molecules for drug-development purposes seeking co-development partners for new small molecules production.
NOVA Inventors
Claúdia Nunes Santos
Diogo Carregosa
Rafael Carecho


